

Our team is proud to present the first fetal monitoring guideline that solely relies on physiology-based interpretation for the assessment of fetal wellbeing. This guideline has been developed by the editorial board based on the experience gained from maternity units where a reduction in the emergency caesarean section rate and/or an improvement in perinatal outcomes was demonstrated after the implementation of physiology-based fetal monitoring.
We would like to take this opportunity to express our gratitude to the fetal wellbeing team and all the maternity staff at St George's Hospital, Lewisham and Greenwich NHS Trust and Kingston Hospital. This guidance is built upon the foundation laid by their collective experiences, contributions and hard work.
We would also like to thank the international consensus panel of expert reviewers from 14 countries, who have embraced a physiological approach to CTG interpretation in their daily clinical practice. We are honoured to have Prof Sir Arulkumaran as a special invited expert reviewer of the physiology-based guideline on CTG interpretation. The editorial board would like to take this opportunity to acknowledge his immense contribution to intrapartum fetal monitoring, and especially, for disseminating the knowledge on fetal physiological response to intrapartum hypoxic stress through several of his publications.
This guideline describes fetal monitoring using physiology-based CTG interpretation. It has been developed by the editorial board based on the experience gained from maternity units where a reduction in the emergency caesarean section rate and/or an improvement in perinatal outcomes was demonstrated after the implementation of physiology-based fetal monitoring.
It is important to stress that fetal monitoring is only part of the overall clinical assessment of both mother and fetus, aimed mainly at the detection of fetal hypoxia. This guidance must be used within the context of the whole clinical picture, taking into account other non-hypoxic factors causing fetal injury. This is particularly important when events are evolving rapidly necessitating interventions irrespective of fetal monitoring.
This guidance is based on the evidence available to the editorial board at the time of creating this document, which are listed in the reference section of this document. We recognise that it is impossible for any guideline to cover every clinical scenario, hence it is important for clinicians using this guidance to apply it in accordance with their clinical expertise and logic, and to seek a second opinion whenever required.
We would like to take this opportunity to express our gratitude to the fetal wellbeing team and all the maternity staff at St George’s Hospital, Lewisham and Greenwich NHS Trust and Kingston Hospital. This guidance is built upon the foundation laid by their collective experiences, contributions and hard work. We dedicate this guideline to help improve the outcome of mothers and babies all over the world.
Lead Consultant Labour Ward and Acute Gynaecology at St. George's University Hospitals NHS Foundation Trust, London
Honorary Senior Lecturer St George's University of London
Fetal wellbeing midwife at Lewisham and Greenwich NHS Trust
Member of the Sign up to Safety Project and co-author of the fetal monitoring guideline at Lewisham and Greenwich NHS Trust
Clinical Fellow in Obstetrics and Gynaecology at St George’s University Hospital NHS Foundation Trust, London
Member of the Sign up to Safety Project and co-author of the fetal monitoring guideline at Lewisham and Greenwich NHS Trust
Consultant Obstetrician and Sub-Specialist in Maternal and Fetal Medicine at Kingston Hospital NHS Foundation Trust, London
Audit and Quality Improvement Lead, Lead Consultant for the Sign up to Safety Project
Senior midwife at Kings College Hospital NHS Foundation Trust
A former member of the Sign up to Safety Project and co-author of the fetal monitoring guideline at Lewisham and Greenwich NHS Trust
Speciality doctor in Obstetrics and Gynaecology at Lewisham and Greenwich NHS Trust, London
Member of the Maternity Transformation project and the Sign up to Safety Project, and co-author of the fetal monitoring guideline at Lewisham and Greenwich NHS Trust
The editorial board wishes to thank the international consensus panel of expert reviewers from 14 countries, who have embraced a physiological approach to CTG interpretation in their daily clinical practice. We are honoured to have Prof Sir Arulkumaran as a special invited expert reviewer of the physiology-based guideline on CTG interpretation. The editorial board would like to take this opportunity to acknowledge his immense contribution to intrapartum fetal monitoring, and especially, for disseminating the knowledge on fetal physiological response to intrapartum hypoxic stress through several of his publications.
This is the first fetal monitoring guideline that solely relies on physiology-based interpretation for the assessment of fetal wellbeing. Previous guidance has been mainly based on pattern recognition. We aim to encompass a pathophysiological approach to explain how a fetus defends itself against intrapartum hypoxic ischaemic insults and highlight the signs that suggest progressive loss of compensation.
The purpose of intrapartum surveillance, in general, is a timely detection of babies who may be hypoxic, so that additional assessments of fetal wellbeing may be used or the baby be delivered by caesarean or instrumental vaginal birth, to prevent perinatal/neonatal morbidity or mortality. NICE 2014, FIGO 2015
As a result of a greater understanding and incorporation of physiology into the interpretation we expect to see a reduction in unnecessary intervention as well as a reduction in fetal hypoxic neurological injury, stillbirth and early neonatal death.
For the reason of simplicity, the editorial board have used the definitions below. These definitions were developed by other professional bodies and guidelines and have been referenced accordingly.
The mean fetal heart rate rounded to increments of five beats per minute during a ten-minute segment, excluding accelerations, deceleration and periods of marked FHR variability. The baseline must be for a minimum of 2 minutes in a ten-minute segment. Otherwise, the baseline for that segment is described as indeterminate.Macones et al. 2008
In tracings with unstable FHR signals, review of previous segments and evaluation of longer time periods may be necessary to determine the baseline. FIGO 2015
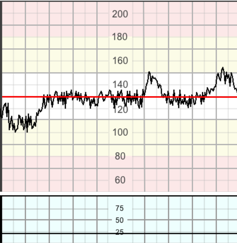
This refers to the oscillation in the FHR signal, evaluated as the average bandwidth amplitude of the signal in 1-minute segments; FIGO 2015 the fluctuations should be irregular in amplitude and frequency. Macones et al. 2008 Variability is documented in beats per minute.
bandwidth amplitude of 5 − 25 bpm.
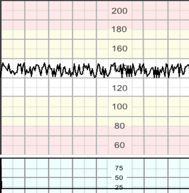
a bandwidth amplitude below 5 bpm for more than 50 minutes in baseline segments, or for more than 3 minutes during decelerations. FIGO 2015, Hamilton et al 2012
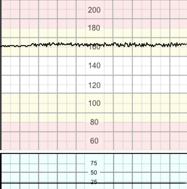
Amplitude range undetectable with or without fetal decelerations. Macones et al. 2008
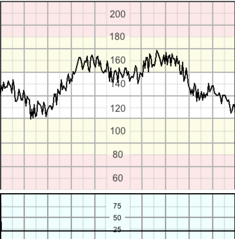
a bandwidth value exceeding 25 bpm lasting more than 30 minutes. The pathophysiology of this pattern is incompletely understood, but it may be seen linked with recurrent decelerations, when hypoxia/acidosis evolves very rapidly. It is presumed to be caused by fetal autonomic instability/hyperactivity. FIGO 2015 Intervention may be required sooner if this pattern is seen during the second stage or during decelerations. A saltatory pattern for more than 30 minutes may indicate hypoxia even without decelerations.
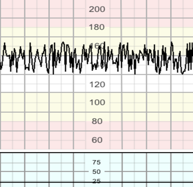
A regular, smooth, undulating signal, resembling a sine wave, with an amplitude of 5−15 bpm, and a frequency of 3−5 cycles per minute. This pattern lasts more than 30 minutes and coincides with absent accelerations.
The pathophysiological basis of the sinusoidal pattern is incompletely understood, but it occurs in association with severe fetal anaemia, as is found in anti-D alloimmunisation, fetal-maternal haemorrhage, twin-to-twin transfusion syndrome, and ruptured vasa praevia. It has also been described in cases of acute fetal hypoxia, infection, cardiac malformations, hydrocephalus, and gastroschisis.FIGO 2015
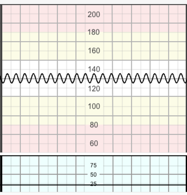
A pattern resembling the sinusoidal pattern, but with a more jagged “saw-tooth” appearance, rather than the smooth sine-wave form. Its duration seldom exceeds 30 minutes and it is characterized by normal patterns before and after. FIGO 2015
Some authorities consider a “pseudo-sinusoidal pattern” as the presence of accelerations with sinusoidal patterns. The presence of “saw toothed” or “Poole shark-teeth” pattern, is termed “atypical sinusoidal pattern” by some authorities, caused by fetal hypotension occurring secondary to acute feto-maternal haemorrhage and conditions such as ruptured vasa praevia. Yanamandra and Chandraharan 2014 This pattern has been described after analgesic administration to the mother, and during periods of fetal sucking and other mouth movements. It is sometimes difficult to distinguish the pseudo-sinusoidal pattern from the true sinusoidal pattern, leaving the short duration of the former as the most important variable to discriminate between the two. FIGO 2015

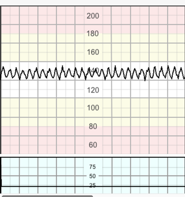
Abrupt (onset to peak in less than 30 seconds) increases in FHR above the baseline, of more than 15 bpm in amplitude, and lasting more than 15 seconds but less than 10 minutes. Before 32 weeks of gestation, amplitude and duration of accelerations may be lower (10 seconds and 10 bpm of amplitude). Macones et al. 2008 An acceleration must start from and return to a stable baseline. GCP
Accelerations coinciding with uterine contractions, especially in the second stage of labour, suggest possible erroneous recording of the maternal heart rate, since the FHR more frequently decelerates with a contraction, while the maternal heart rate typically increases. Nurani et al 2012
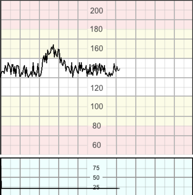
Decreases in the FHR below the baseline, of more than 15 bpm in amplitude, and lasting more than 15 seconds. Decelerations are considered to be a reflex response to protect the myocardial workload when a fetus is exposed to a hypoxic or a mechanical stress, to help maintain an aerobic metabolism within the myocardium.
Decelerations that are gradual (onset to nadir ≥30s) that return to the baseline. They coincide with contractions, Macones et al. 2008 and show normal variability within the deceleration. They are likely to be seen in the late first stage and second stage of labour and are believed to be caused by fetal head compression. They do not indicate fetal hypoxia/acidosis. FIGO 2015
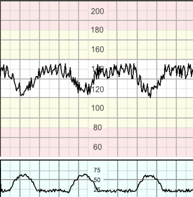
V-shaped decelerations that exhibit a rapid drop (onset to nadir in <30s) followed by a rapid recovery to the baseline. The precipitous fall and rise of the baseline due to cord compression means there is not time to exhibit good variability within the trough of the deceleration. These decelerations vary in size, shape, and relationship to uterine contractions.
Variable decelerations constitute the majority of decelerations during labour, and they translate a baroreceptor-mediated response to increased arterial pressure, as occurs with umbilical cord compression. FIGO 2015 They are believed to occur secondary to baro-receptor and/or peripheral chemo-receptor stimulation. They are seldom associated with fetal hypoxia/acidosis, unless they evolve to exhibit a U-shaped component (“sixties” criteria) with a reduced or an increased variability within the deceleration (see late decelerations below), and/or their individual duration exceeds 3 minutes FIGO 2015, Hamilton et al 2012 (see prolonged decelerations below).
Variable decelerations meet the “sixties” criteria if two or more of the following are present: drops by 60bpm or more, reaches 60bpm or less, for the duration of 60 seconds or longer. Hamilton et al 2012
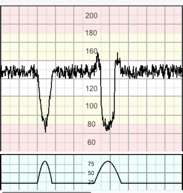
Decelerations with a gradual onset and/or a gradual return to the baseline and/or reduced or increased variability within the deceleration. Gradual onset and return occurs when more than 30 seconds elapses between the beginning/end of a deceleration and its nadir. When contractions are adequately monitored, late decelerations start more than 20 seconds after the onset of a contraction, have a nadir after the acme, and a return to the baseline after the end of the contraction. FIGO 2015
These decelerations are indicative of a chemoreceptor-mediated response to fetal hypoxaemia. Hamilton et al 2012 In a trace showing no accelerations and reduced variability, the definition of late decelerations also includes those with an amplitude of 10−15 bpm (shallow decelerations).
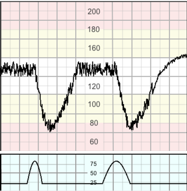
Decelerations lasting more than 3 minutes. These are likely to include a chemoreceptor-mediated component and thus to indicate hypoxemia. Decelerations exceeding 5 minutes, with FHR maintained at less than 80 bpm and reduced variability within the deceleration, are frequently associated with acute fetal hypoxia/acidosis and require urgent intervention FIGO 2015 (see “3 – minute rule”).
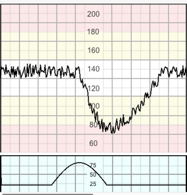
These are recorded as bell-shaped gradual increases in the uterine activity signal followed by roughly symmetrical decreases. With the tocodynamometer, only the frequency of contractions can be reliably evaluated. FIGO 2015 The intensity and duration of contractions may be assessed by manual palpation. If frequency of contractions cannot be assessed reliably by the tocodynamometer, manual palpation for 10 minutes every 30 minutes is required. GCP
This refers to periods of:
The alternation of different behavioural states (cycling) is a hallmark of fetal neurological responsiveness and absence of hypoxia/acidosis. Transitions between the different patterns become clearer after 32−34 weeks of gestation, consequent to fetal nervous system maturation.
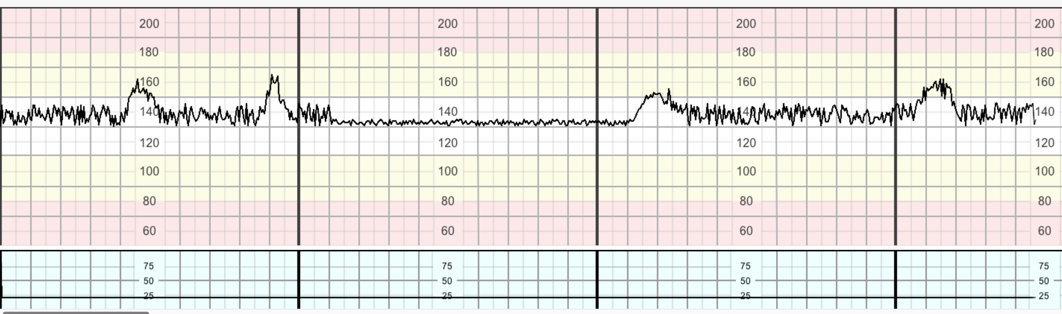
During labour the fetus employs various adaptive mechanisms in response to hypoxia, these generally follow a similar pathway as the physiological response to exercise. Intrapartum hypoxia generally follows one of three pathways:
Presents as a prolonged deceleration lasting for more than 5 minutes or for more than 3 minutes if associated with reduced variability within the deceleration. FIGO 2015
3 Accidents
2 Iatrogenic causes
Fetal pH drops at a rate of 0.01/min during the deceleration Gull et al. 1996
Management follows the 3-Minute Rule:
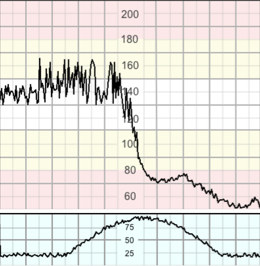
Presents on the CTG by the fetus spending most of the time in decelerations.
This is almost invariably caused by uterine hyperstimulation.
Fetal pH drops at a rate of 0.01 / 2-3 minutes
Management is by
If encountered in the second stage of labour, ask the mother to stop pushing to allow the recovery of the fetal status. If no improvement is seen within 10 minutes, expedite delivery. Once stable recommence pushing. If subacute hypoxia recurs, expedite delivery.
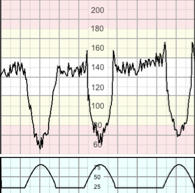
This is the most common type of hypoxia in labour
During this process, the fetus undergoes the same changes that a normal adult would be expected to show during exercise
Management of gradually progressive hypoxia is by improving fetal conditions with the first signs of redistribution to avoid internal organ damage. (stage 4).
Fetal pH drops at a rate of 0.01 / 2-3 minutes
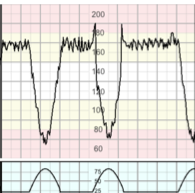
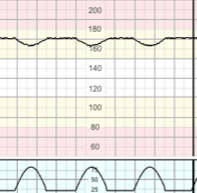
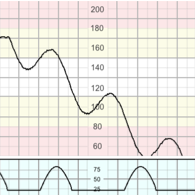
(This is an antenatal type of hypoxia with implications for intrapartum care)
Presents as a baseline rate at the upper end of normal associated with reduced variability and blunted responses (infrequent accelerations and lack of cycling) and is frequently associated with shallow decelerations.
This represents a fetus with reduced reserve and increased susceptibility to hypoxic injury during labour.
Careful consideration should be given when planning interventions potentially increasing the risk of hypoxia, with low threshold for surgical intervention.
A useful checklist that helps exclude signs of chronic hypoxia is presented below (see table 3).
All women that have existing medical or obstetric conditions should have an obstetric review during pregnancy with a full plan of care formulated for labour and birth. This should include the suitability for different birth settings and the type of fetal monitoring required when in labour. The care plan should be explained to and agreed by the woman.
Continuous electronic fetal monitoring (CEFM) in low risk women is associated with an increased level of intervention without any improvement in outcome. Maude et al 2014 Women who are healthy and have had an uncomplicated pregnancy should be offered and recommended intermittent auscultation to monitor fetal well-being. This should be performed using a Doppler ultrasound or pinard stethoscope. NICE 2014 A woman must be fully informed of the risks and benefits of intermittent auscultation (IA) and CEFM. If during labour, she chooses not to be monitored by the recommended method a full discussion of the potential impact on her and the fetus should be undertaken and the labour ward coordinator and senior obstetrician should be informed. This discussion must be clearly documented in the patient’s records.
There does not appear to be any good evidence from trials to recommend any particular frequency and duration of IA. Therefore, it is more a ‘custom of practice’ than ‘evidence-based approach’. Walsh 2008 Auscultation is carried out every 15 minutes in the first stage and every 5 minutes in the second stage - a practice adopted from the randomized controlled trials comparing IA and CEFM. On assessing the woman and establishing that she is low risk and is suitable for IA the method is as follows:
During the course of pregnancy or labour the clinical circumstances may change, increasing risk to mother and/or fetus (see table-1). In this situation, the mother should be informed of the rationale for changing the method of auscultation and should also be clearly documented in the notes.
If CEFM has been commenced due to concerns arising during IA but the CTG is normal after a minimum of 30 minutes, it is deemed suitable to return to IA. Maude et al 2014 If concerns arise again, CEFM would be recommended until delivery.
If conversion to CEFM is advised but declined, the risks of not continuously monitoring should be explained, and the midwife in charge and obstetric team informed. All discussions must be clearly documented in the notes.
| Table 1 – Risk factors indicating conversion from Intermittent Auscultation to Continuous Electronic Fetal Monitoring | |
|---|---|
| Maternal | Fetal |
| Pulse over 120 beats/minute on 2 occasions 30 minutes apart * | Undiagnosed breech presentation; transverse or oblique lie (review mode of delivery) |
| A single reading of diastolic blood pressure ≥ 110 mmHg or systolic blood pressure ≥ 160 mmHg * | Free-floating head in a nulliparous woman |
| Diastolic blood pressure 90 to 109 mmHg or systolic blood pressure of 140 to 159 mmHg on 2 consecutive readings taken 30 minutes apart * | Recurrent Accelerations (immediately following a contraction i.e. overshoot) |
| Maternal pyrexia (defined as 38.0 °C once or 37.5 °C on two occasions 1 hour apart) | Fetal heart rate below 110 or above 160 beats/ minute, or if it is perceived as inappropriate for gestational age. |
| Any vaginal blood loss other than a show | Evidence of a rising baseline on the partogram |
| The presence of meconium if birth is not imminent NICE 2014 | 2 x decelerations in fetal heart rate heard on intermittent auscultation after 2 successive contractions |
| Persistent pain in between contractions | |
| Epidural analgesia | |
CEFM could potentially reduce mobility. However, every effort should be made to facilitate the normal physiology of labour by encouraging the woman to adopt upright positions and mobilise. This can be facilitated by the use of wireless telemetry or the encouragement to move within the constraints of being connected to the monitor.
CEFM is a screening tool for hypoxia and does not replace the need for accurate clinical observations on which decisions should be made in conjunction with the CTG. FIGO 2015
The table below is not exhaustive, any condition which is thought to increase the risk of fetal hypoxia mandates CEFM.
| Table 2 – Inclusion criteria for Continuous Electronic Fetal Monitoring | |
|---|---|
| Maternal indication | Fetal indication |
| Gestation <37 or >42 weeks | Abnormal Doppler artery velocimetry |
| Induced labour | Known or suspected IUGR |
| Administration of oxytocin | Oligohydramnios or polyhydramnios |
| Ante/Intrapartum haemorrhage | Malpresentation |
| Maternal illness (e.g. diabetes, cardiac, renal, hyperthyroidism). * | Meconium stained liquor |
| Pre-eclampsia | Multiple pregnancy (all babies to be monitored) |
| Previous uterine scar (caesarean section or myomectomy) | Suspected small for gestational age or macrosomia |
| Contractions < 5:10 or lasting for more than 90 seconds | Reduced fetal movements in the last 24 hours reported by the woman |
| During / following insertion of an epidural block | Two-vessel cord |
| Prolonged rupture of membranes > 24 hours unless delivery is imminent. | A rise in baseline, repeated decelerations or slow to recover decelerations, or overshoots |
| Maternal request | Fetal structural abnormalities diagnosed during the antenatal period and planned for CEFM |
| Physiological Classification of Hypoxia | ||
|---|---|---|
| Hypoxia | Features | Management |
| No Hypoxia |
|
|
| Evidence of Hypoxia | ||
| Chronic Hypoxia |
|
|
| Gradually Evolving Hypoxia | Compensated |
|
| Rise in the baseline (with normal variability and stable baseline) preceded by decelerations and loss of accelerations | ||
| Decompensated |
|
|
|
||
| Subacute Hypoxia |
|
First Stage |
|
||
| Second Stage | ||
|
||
| Acute Hypoxia | Prolonged Deceleration (> 3 minutes) | Preceded by reduced variability and lack of cycling or reduced variability within the first 3 minutes |
| Immediate delivery by the safest and quickest route | ||
| Preceded by normal variability and cycling and normal variability during the first 3 minutes of the deceleration | ||
|
||
| Unable to Ascertain fetal wellbeing | (Poor signal quality, uncertain baseline, possible recording of the maternal heart rate) |
|
| Table 4 - CTG Assessment Tool | |||||||
|---|---|---|---|---|---|---|---|
| Baseline | bpm | Variability | bpm | Accelerations | Decelerations | ||
| Rise in Baseline (≥ 10%) | Yes | No | |||||
| Inter-contraction interval > 90 sec | Yes | No | |||||
| Maintained Cycling | Yes | No | |||||
| Abnormal Variability (<5 or >25) | Yes | No | |||||
| Features of Hypoxia | Yes | No | |||||
| Type: | |||||||
| Central Organs well oxygenated | Yes | No | |||||
| Other risk factors noted | |||||||
| Recommended Management | |||||||
Identify reversible causes as alleviating them can lead to subsequent recovery of adequate fetal oxygenation and the return to a normal trace.
When CTG changes develop, it is important to address underlying causes before hypoxia occurs. The midwife caring for the woman should escalate to a senior midwife/obstetric team for review without delay.
CTG must be stored for 25 years. Given that thermal paper deteriorates and is only legible for about 10 years, storage should ideally be in electronic form.
Be aware that if the CTG parameters of baseline fetal heart rate and baseline variability are stable, the risk of fetal acidosis is low. NICE 2014, FIGO 2015
It is important to attempt to understand the physiological events behind changes in fetal heart rate pattern (see above). This can provide reassurance of fetal status without the need to perform further testing. However, in situations where changes cannot be explained, it is important to seek senior advice and plan further testing accordingly.
There are many observational studies supporting the use of fetal scalp stimulation (FSS) compared to fetal blood sampling. However, the evidence grading for the trials behind the use of FSS is predominantly moderate to low. Skupski et al 2002
There is no consensus on the clinical situation for FSS to be used. FIGO 2015
FSS involves stimulating the fetal scalp by rubbing it with the examiner’s fingers. Other techniques involve using forceps to clasp the fetal skin, or alternatively using vibroacoustic stimulation applied to the mother’s abdomen. However, these are not locally available. Elimian 1997 Digital scalp stimulation is the most widely used as it is the easiest to perform, least invasive, and appears to have a similar predictive value for fetal hypoxia/acidosis to the other alternatives. FIGO 2015
If an acceleration is noted at FSS the likelihood of fetal hypoxia is <2.5%, while in the absence of an acceleration, the likelihood of fetal hypoxia is >38%. Skupski et al 2002 The risk of hypoxia is increased if the lack of acceleration is associated with reduced variability. Elimian 1997 The information should be considered in the context of the entire clinical picture. NICE 2014, FIGO 2015
There have been six meta-analyses assessing the effectiveness of STAN in ascertainment of fetal wellbeing. Five of these have shown that although STAN reduces the need for fetal scalp blood sampling and operative vaginal delivery, there was no improvement in number of births by caesarean section, babies with low APGAR scores at 5 minutes, severe fetal metabolic acidosis, or the number of babies with neonatal encephalopathy. Adalina et al 2015 The sixth meta-analysis claimed to have found a mistake within the other five, and after correcting for this, it showed reduction of rates of metabolic acidosis. Olofsson et al 2014 The latest meta-analysis which included the largest US randomised trial on STAN Blix et al, 2016 has reported 36% statistically significant reduction in neonatal metabolic acidosis and 8% reduction in operative vaginal birth. Therefore, in the opinion of the authors, STAN remains the only additional test of fetal wellbeing with robust scientific evidence. Bhide et al, 2016, Chandraharan E, 2018.
It is a combined assessment of the standard fetal heart rate trace with an automated analysis of the fetal electrocardiogram. This is obtained by placing a spiral electrode on the fetal scalp. The machine initially analyses the ST segments (required 20 ‘x’s) to obtain a baseline shape of the ECG complex, which usually takes the first 4 minutes. Subsequently, it compares the complex every 30 beats for changes indicating the presence of possible ischaemia. With every successful comparison made, an ‘x’ is indicated at the bottom of the trace.
Trace interpretation needs to take into account the CTG pattern classification and the degree of ST changes. The system’s automatic warnings of ST events occur when it detects changes in ECG morphology when compared with the previous state. (table 5). A Flow Chart to aid management when using STAN for fetal monitoring is provided.
| Table 5 - Classification of CTG | |||
|---|---|---|---|
| Baseline Rate | Variability and Reactivity | Decelerations | |
| Normal CTG |
|
|
|
| Intermediary CTG |
|
|
|
| The combination of several intermediary factors results in an abnormal CTG | |||
| Abnormal |
|
|
|
| Pre-terminal | Total lack of variability (<2bpm) and reactivity with or without decelerations | ||
| ST Analysis | ||||
|---|---|---|---|---|
| ST Event | Normal CTG | Intermediary | Abnormal | Pre-terminal |
| Episodic T-QRS rise |
|
> 0.15 | > 0.10 | Immediate delivery |
| Baseline T-QRS rise | > 0.10 | > 0.05 | ||
| Biphasic ST | 3 biphasic log messages | 2 biphasic log messages | ||
The above guideline is based on FIGO CTG Guidelines of 1987 as ST-Analyser was validated using these guidelines. Based on the experience and perinatal outcomes of the maternity units which have implemented STAN after training staff on fetal physiology, the Editorial Board recommends the use of the STAN guideline in combination of deeper understanding of fetal physiology.
The Cochrane systematic review in 2013 has demonstrated that there is no available evidence of a correlation between fetal scalp pH and improvement in long term outcomes. In addition, the review has also demonstrated that contrary to the erroneous belief in the past, current evidence suggests that FBS may increase the number of caesarean sections and operative vaginal births. Further review of evidence has shown rare, but, potentially serious fetal complications. Therefore, as current scientific evidence does not support the use of FBS in clinical practice as its benefits no longer outweigh its risks, the authors of this guideline do not support the use of FBS as an adjunctive technique for the assessment of fetal well-being.
Other factors that are present during labour such as prolonged rupture of membranes, defined as spontaneous rupture of membranes for greater than 24 hours, chorioamnionitis, anhydramnious, meconium-stained liquor, maternal infection or pyrexia, and the speed of evolution of hypoxia are likely to modify the responses of the fetus as well as affect the perinatal outcome. Sacco et al 2015
Meconium stained liquor (MSL) can be present in a normal post term fetus without an indication that the baby has experienced hypoxia. In a preterm fetus, <34/40, the presence of meconium signifies that there is likely infection, such as listeria, ureaplasma or rotavirus. Blot et al. 1983 Clear liquor has antibacterial properties, however in the presence of meconium these properties are restricted. Unsworth and Vause 2010 With thick meconium, E coli has the ability to grow rapidly, whereas Group B Streptococcus proliferates even in clear liquor. Eidelman et al. 2002 Fetal tachycardia (≥160 bpm), in the presence of MSL has a Relative Risk of 51 for the development of chorioamnionitis, in comparison to clear liquor. Blot et al. 1983
MSL is associated with complications in the newborn. The most severe complication is meconium aspiration syndrome (MAS). Unsworth and Vause 2010 Aspiration of meconium can occur in-utero with fetal gasping, or after birth, with the first breaths of life. Mundhra and Agarwal 2013
There is still no effective and safe treatment or prophylactic measure for MAS once the meconium has passed below the vocal cords into the lungs. Chandraharan and McDonnell 2015
Evidence shows that when the placental oxygen supply is interrupted, the fetus attempts to breathe. Should these attempts fail to provide an alternative oxygen supply, and if hypoxia continues, the respiratory centre becomes unable to continue initiating breathing and the breathing stops, usually within 2 to 3 minutes. MOET 2014
In view of this, extra vigilance should be taken to observe for signs of hypoxia in the presence of meconium. A lower threshold for expediting delivery should be considered when there is meconium and signs of hypoxia as a CTG cannot predict if a fetus will gasp or when this would happen.
If a fetus has passed meconium, the mother should be informed that there is a risk of meconium already being present in the lungs. Most meconium will be expelled from the fetal lungs as the baby passes down the birth canal but in 1-3% of live births, the baby will develop MAS. Impey et al. 2008
Care should be taken when using prostaglandins or oxytocin to augment or stimulate labour. One of the iatrogenic causes of prolonged decelerations includes prolonged or frequent uterine contractions secondary to oxytocin. If this cause is identified, immediate action should be taken to improve utero-placental oxygenation by stopping oxytocin and changing maternal position to reduce the stress the baby is experiencing. FIGO 2015 Consideration should also be given to starting acute tocolysis using a beta-adrenergic agonist such as terbutaline. NICE 2014
CEFM is necessary with oxytocin augmentation. If the fetal heart rate is normal, oxytocin should be titrated to achieve contractions at a rate of 4:10. It should be reduced if contractions occur more frequently than 5:10. If evidence/suspicion of fetal decompensation occur, oxytocin infusion should be stopped and an urgent assessment of the fetal condition should be undertaken and documented by an obstetrician. Arulkumaran et al. 2004 In the event of acute hypoxia, oxytocin should be stopped, and the 3-minute rule initiated. A full assessment of the fetal condition must be undertaken and documented by an obstetrician BEFORE oxytocin is recommenced.
Heat transmission during pregnancy results in fetal temperature being 0.3-0.5°C higher than maternal temperature. The umbilical circulation transfers 85% of the heat produced by the fetus to the maternal circulation. The remaining 15% is dissipated through the fetal skin to the amnion and is then transferred through the uterine wall to the maternal abdomen. Lieberman et al. 2000 If there is pyrexia, the metabolic demands of the fetal tissues are increased and so the risk of hypoxia is elevated. Holt et al. 1994 This should be considered especially when using oxytocin, and a prolonged labour should be avoided. The combination of maternal pyrexia with cord acidosis [indicative of fetal acidosis] greatly increases the risk of neonatal encephalopathy. Evidence suggests that acidosis and pyrexia represent two separate causal pathways of neonatal encephalopathy leading to a cumulative effect. Impey et al. 2008
There is no clear evidence to suggest when a fetus should be delivered if there is maternal or fetal infection. In view of the lack of clear evidence/guidance on a safely acceptable time frame for delivery, a clear discussion with the mother should be undertaken with agreed management plan and time frame documented from this. Measures, such as paracetamol, IV fluids and IV antibiotics, should be used to treat any pyrexia and infection. Evidence shows that an intrapartum maternal dose of 1500mg of cefuroxime IV produces effective fetal concentrations for prophylaxis, but not treatment. Holte et al. 2004
Intrapartum fever, even when unlikely to be caused by infection, is associated with a fourfold increase in the risk of unexplained, early-onset seizures in term infants. Holt et al. 1994
Major placental abruption is one of the 3 major intrapartum accidents and may present as a single and sudden drop in the baseline rate (acute hypoxia). In this case, delivery must be expedited as it is most likely to be the evidence of a placental abruption and is irreversible. FIGO 2015 It is also important to note that the use of tocolytics in APH may aggravate placental separation causing worsening fetal hypoxia.
This can cause a sudden drop in maternal blood pressure which causes redistribution of maternal blood away from the placenta resulting in inadequate placental perfusion. It will present as a single and sudden drop in the baseline rate (acute hypoxia). In this instance, it is reversible and should be corrected by changing the maternal position and IV fluids ± I.V. ephedrine (to be administered by the anaesthetic team). Greenwell et al. 2012 This vasodilation can also cause an increase in maternal temperature as a result of altered thermoregulation. RCOG 2015
If a woman has had a previous lower segment caesarean section and begins to labour, the risk of uterine scar rupture is between 0.07% Nahum and Isaac 2016 – 0.5% RCOG 2015 and must be considered. This is the third major intrapartum accident and may present as a single and sudden drop in the baseline rate (acute hypoxia). In this case, delivery must be expedited as it is irreversible. FIGO 2015
Research has shown that only 8-12% of women with histologically confirmed chorioamnionitis would demonstrate tachycardia and pyrexia during labour. Therefore, any increase in the baseline fetal heart rate without preceding decelerations should arouse the suspicion of an ongoing subclinical chorioamnionitis. Other clinical parameters, such as presence of meconium, rate of progress of labour, history of prolonged rupture of membranes or prolonged labour and absence of cycling should be considered whilst making management decisions.
There is paucity of evidence/ guidelines on the use of CTG in Preterm babies. This has resulted in some authors advising against continuous monitoring in extreme prematurity (24 – 28 weeks). The key factors affecting FHR characteristics in the preterm fetus are immaturity of the central and peripheral nervous systems, reduced placental reserve, immature adrenal gland and myocardium, and reduced amount of Wharton’s jelly in the umbilical cord.
CTG findings include:
It is important to consider the effect of any medication administered to the mother during labour and anticipate the changes it may cause on the CTG trace. This is even more important when medications are given for the purpose of improving fetal conditions. In such cases, we would need to consider what to look for as signs of improvement, what may occur if our intervention did not work or if the situation is worsening, how soon to expect changes and how long should they last for.